by Dennis Crouch
On March 25, 2026, the House Judiciary Subcommittee on Courts, Intellectual Property, Artificial Intelligence, and the Internet held its first oversight hearing of the USPTO under Director John Squires. The hearing, which ran for several hours with Squires as the sole witness, covered an extraordinary range of topics: IPR institution policy (including the proposed NPRM), retroactive de-institution of trials, AI in patent examination, the application backlog, national security concerns about foreign petitioners, Section 101 reform, fee-setting authority, injunctive relief, employee morale, and a sustained exchange over Dir. Squires decision to file trademark applications for the Trump administration’s “Board of Peace.” Overall, there was substantial and bipartisan skepticism.
Several themes recurred throughout the hearing. Squires repeatedly invoked his “born strong” framework for patent quality and characterized the IPR changes as restoring “balance and fairness” to the AIA system. Members on both sides pushed back, questioning whether the Director’s centralized control of institution decisions, combined with summary denial orders that provide no reasoning, is consistent with the AIA’s design. The hearing also showcased Squires’ rhetorical style: the “Central Bank of Innovation” metaphor appeared in both his written and oral testimony, and he introduced a new characterization of the proposed estoppel rule as “one, join, and done” rather than the critics’ shorthand of “one and done.”
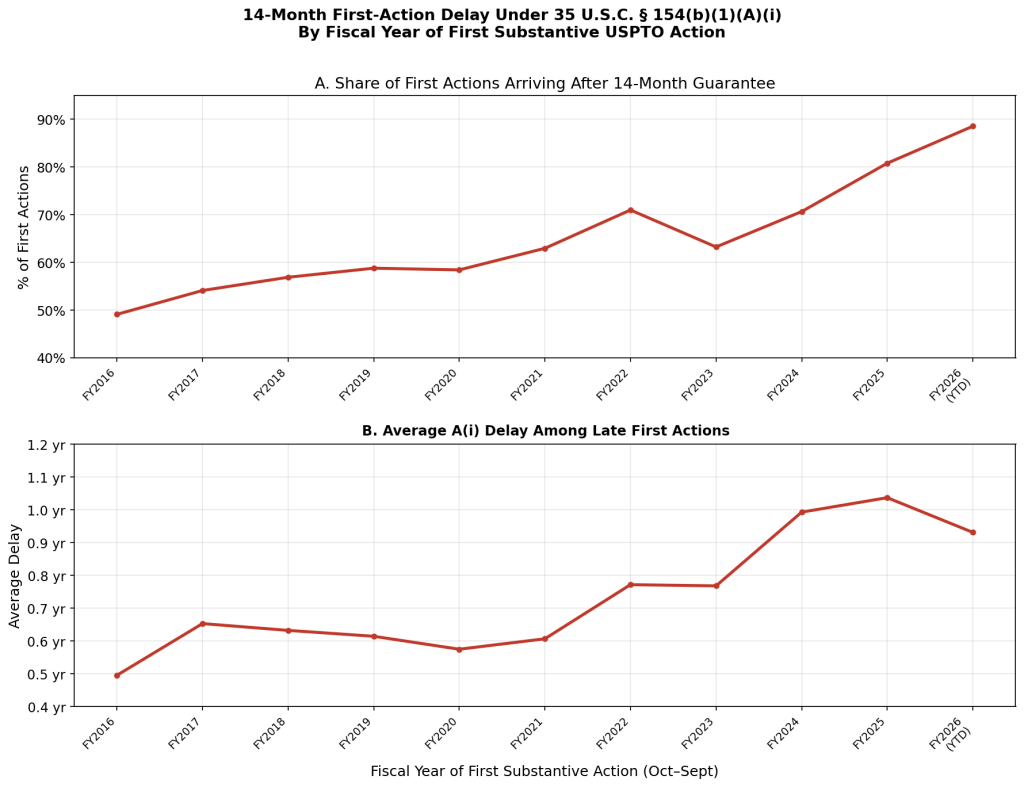
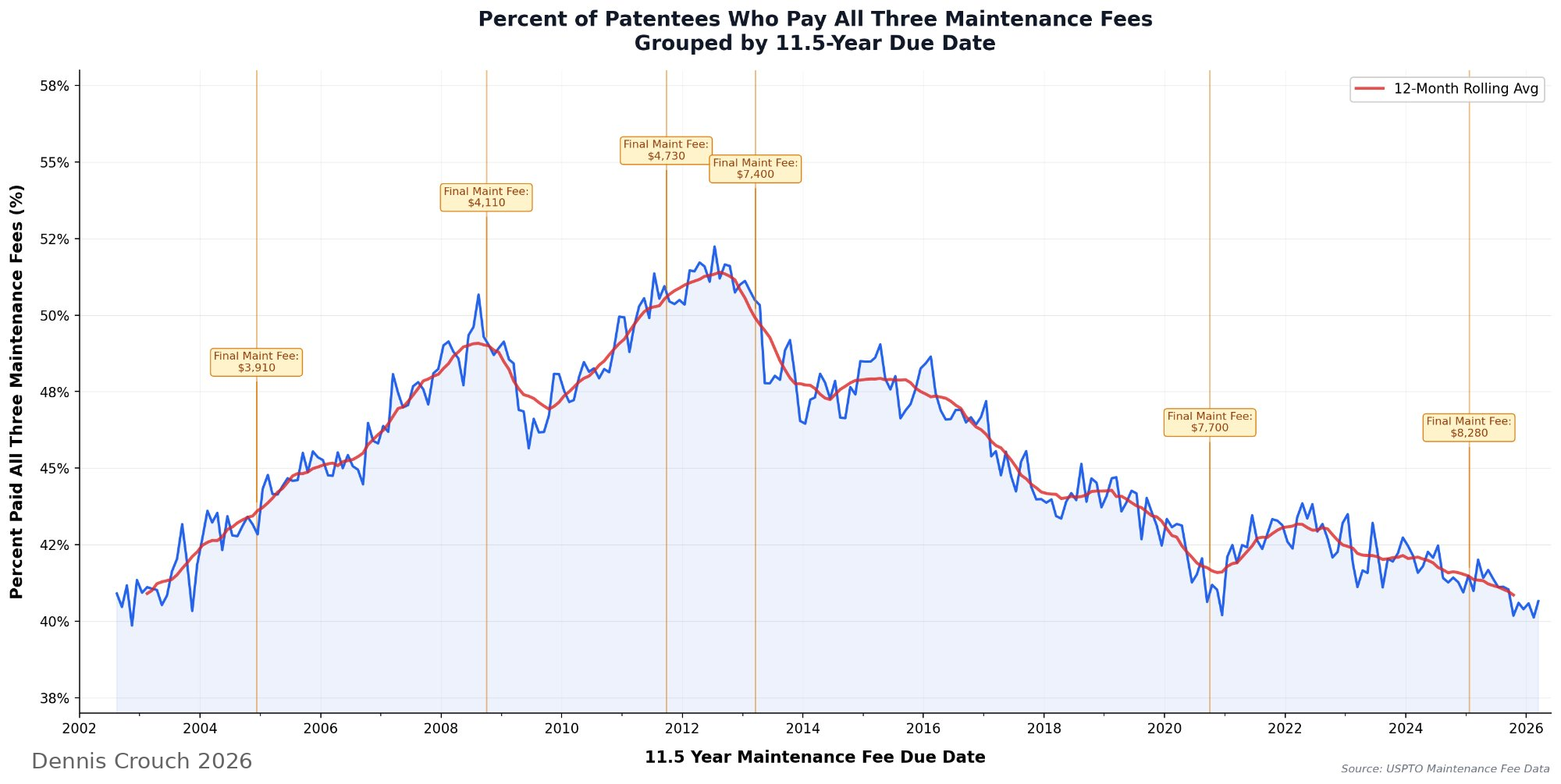
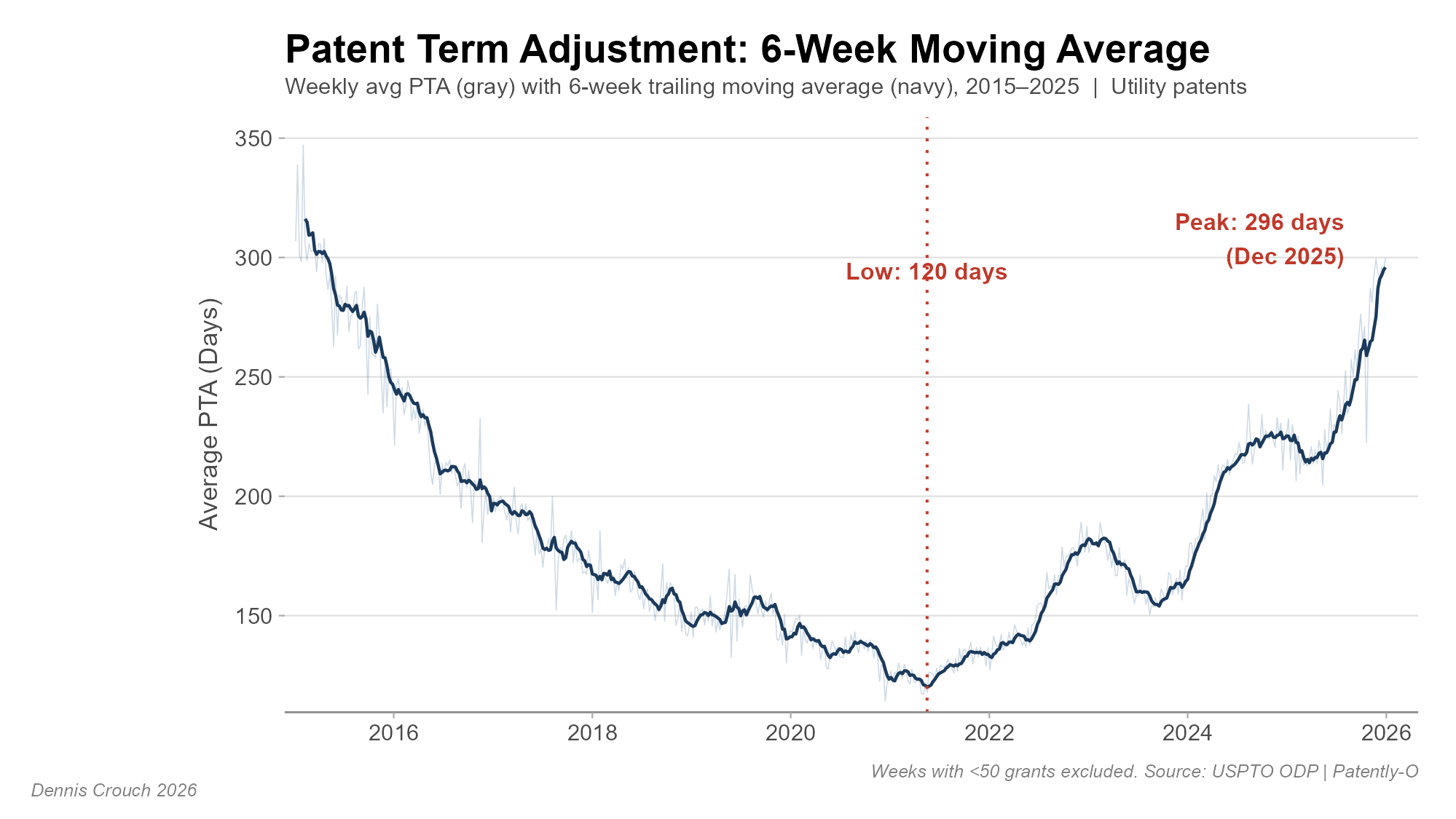
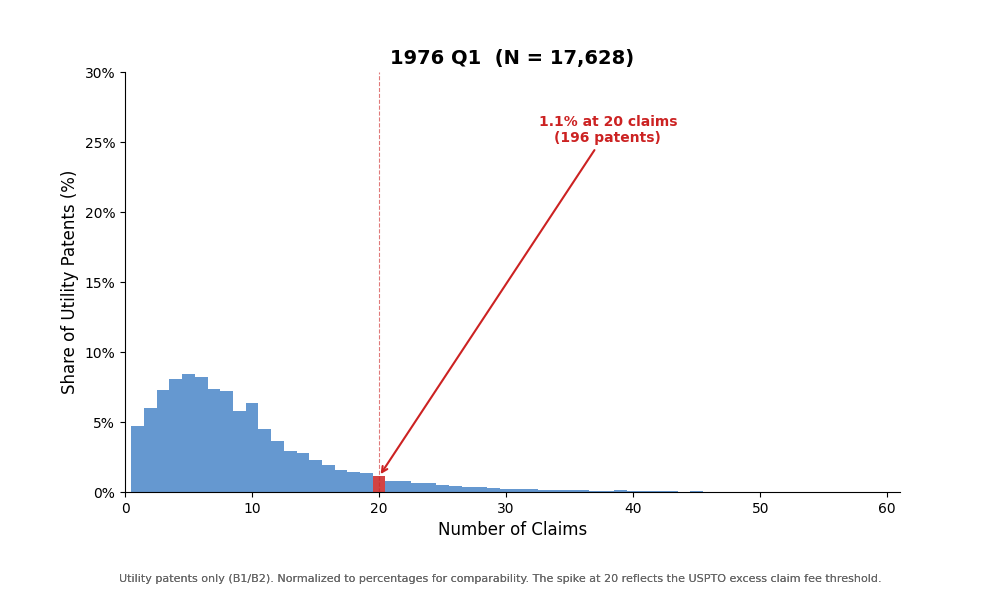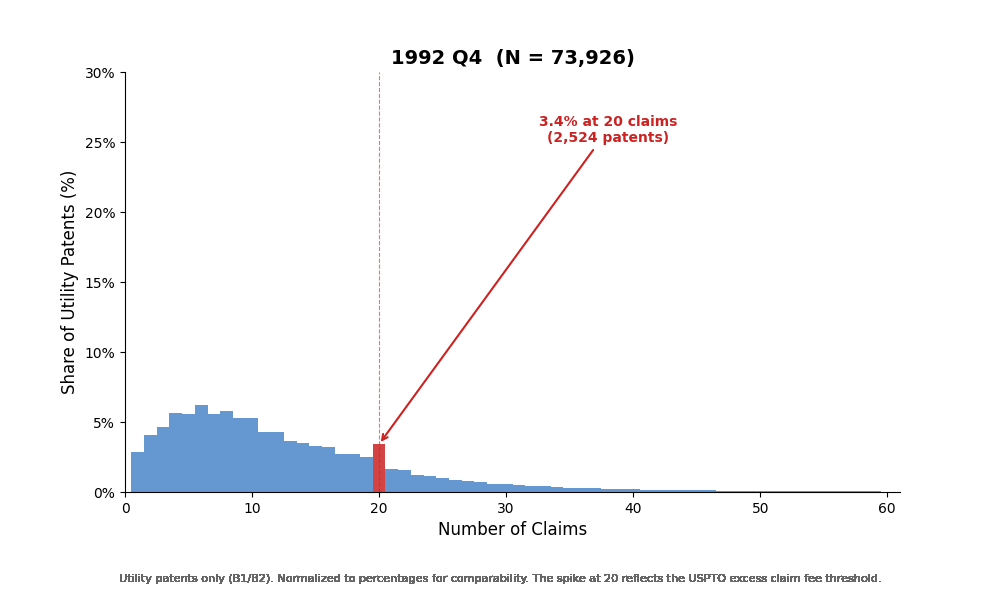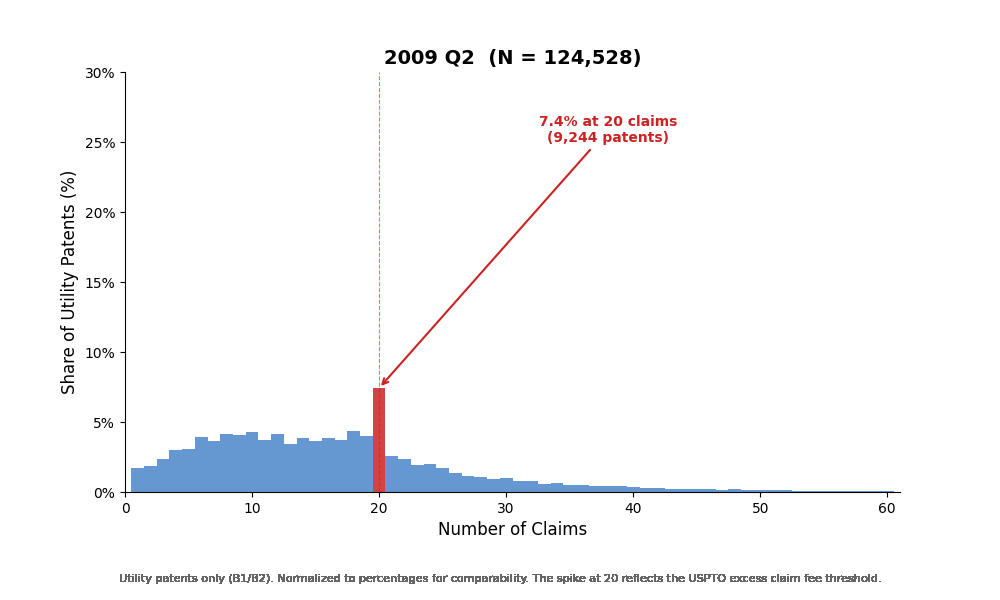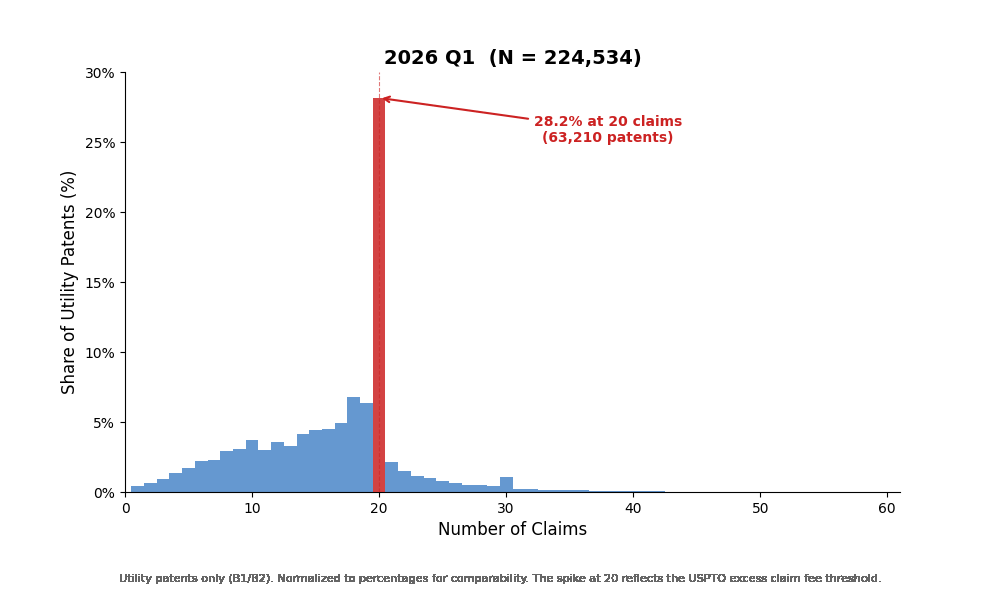
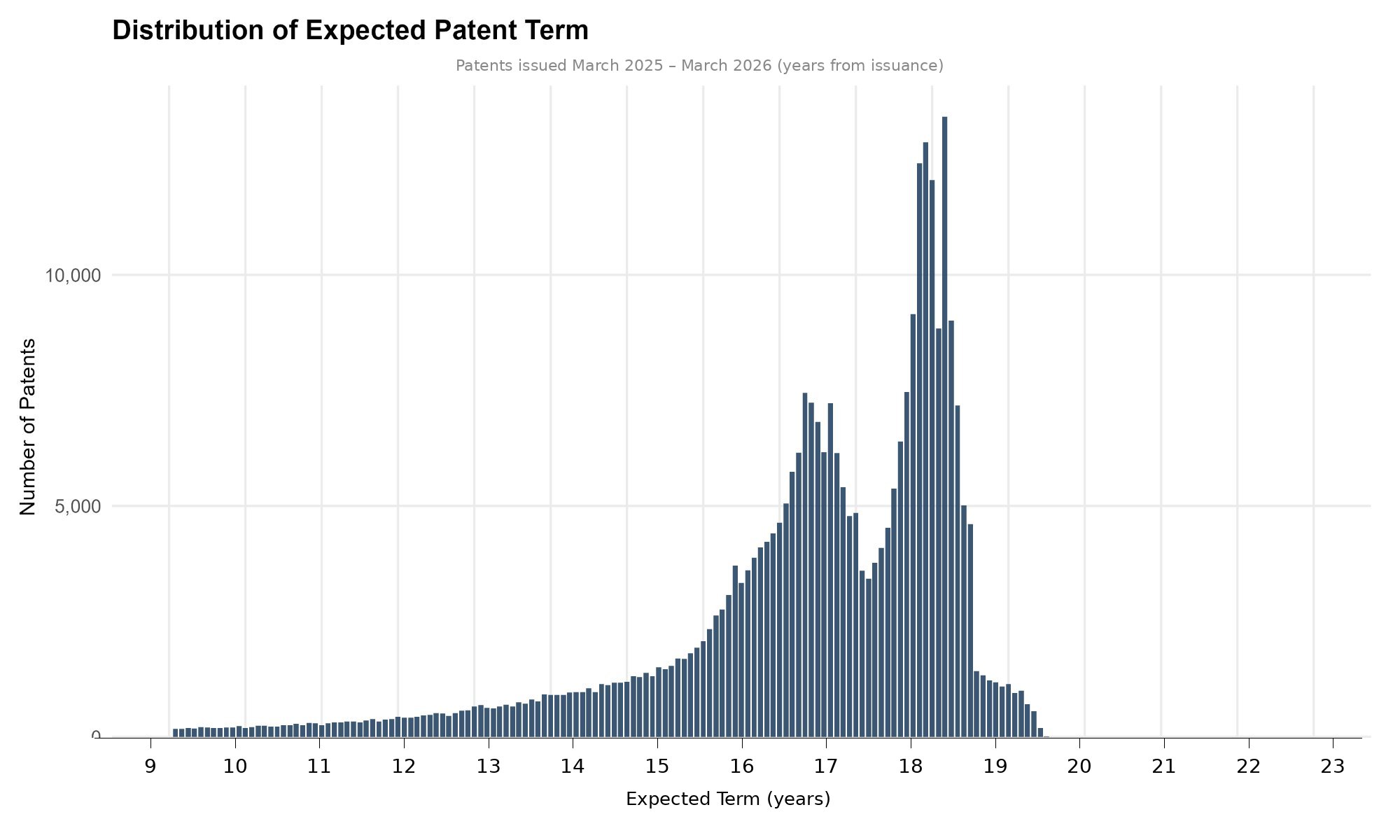 The most striking feature is
The most striking feature is