 Apotex v. Pfizer (Fed. Cir. 2005) (NONPRECEDENTIAL)
Apotex v. Pfizer (Fed. Cir. 2005) (NONPRECEDENTIAL)
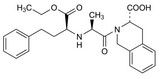
Apotex filed an ANDA to begin manufacturing a generic version of Pfizer’s patented drug quinapril. After Pfizer refused to file an infringement suit, Apotex filed a declaratory judgment action — hoping to prove that the patent was invalid.
At the district court level, this case fell in line with the recent CAFC decision Pfizer v. Teva holding that an ANDA filing does not, by itself, create a reasonable apprehension of suit. In dissent in Teva, Judge Mayer argued that the statute does provide that an ANDA filing constitutes an act of infringement sufficient to trigger a justiciable controversy. As it turns out, Judge Mayer was also a panel member for the Apotex v. Pfizer appeal, along with Judges Plager and Gajarsa.
Less than one week before oral argument, and after learning that Judge Mayer would be a panel member, Pfizer covenanted not to sue Apotex for infringement of U.S. Patent No. 4,743,450. In their decision, the Appellate Panel unanimously found that the covenant not to sue was determinative:
A covenant not to sue, such as that provided by Pfizer, moots an action for declaratory judgment. . . . As a result, the judgment and opinion of the district court are vacated and the case is remanded with instructions to dismiss for lack of jurisdiction.
During oral arguments, the Judges were clearly displeased with Pfizer’s litigation strategy. Although Pfizer won the day, the court did award costs to Apotex.
Links: