Mayo v. Prometheus Labs., (on petition for writ of certiorari 2009)
The Mayo Clinic has filed a petition asking the Supreme Court to hear its case challenging the patentable subject matter of Prometheus Labs’ patents that cover a method of optimizing the dosing of a drug. U.S. Patents 6,355,623 and 6,680,302.
The invention basically offers an iterative approach to dosing that involves three steps: (1) administer a drug to a subject; (2) then determine the level of drug in the subject; and (3) decide whether the next dose should be the same, higher, or lower. The decision on the next dose is made by comparing the the level of the drug in the subject against predetermined thresholds. Claim 1 of the ‘302 patent adds additional detail including identifying the active ingredient (6-thioguanine), the diagnosis (a GI disorder), and the predetermined thresholds (e.g., 230 pmol of drug per 8108 red blood cells). Claim 46 of the ‘623 is a broad claim in that it does not not require the administration step (step 1 from above).
Broad Functional Language: You’ll note that the claims are written in broad functional language without tying the the claimed methods to any particular technology. Thus, the “administering” step could potentially be accomplished by any effective method (that has or will be invented) of getting the drug into the subject. Likewise, the patent offers potential methods of “determining the level” of drug in the body (e.g., liquid chromatography) but the claim is broadly written to seemingly cover any mechanism that fulfills that method.
Novelty Involves the Mental Step: Of course, the general iterative process is well known, what makes the Prometheus claims novel is that they identify the particular thresholds that are important (e.g., 230 pmol). In practice, the process of comparing the thresholds to the subject’s drug level is done in the mind of a physician. As Mayo explains, this merely involves the physician’s mental recognition of a natural correlation between metabolite levels and patient condition.
With these facts in mind, Mayo presents the following question: Whether 35 U.S.C. § 101 is satisfied by a patent claim that covers observed correlations between patient test results and patient health, so that the claim effectively preempts all uses of these naturally occurring correlations.
The Federal Circuit held the method patentable under its Bilski test by finding that the required administration of the drug transformed an article into a different state or thing — essentially creating a per se rule that a method requiring administration of a drug will be deemed patentable subject matter under Section 101. The court also held that the “determining the level” step was necessarily transformative since “those levels cannot be determined by mere inspection.”
Chances for Supreme Court Review: The fact that Bilski – another Section 101 case – is already pending before the Supreme Court does not mean that the court will reject this case. Rather, the court has often granted certiorari in tandem. The Supreme Court has regularly used case clustering in the past when considering patent issues: Nonobviousness (John Deere (1966), Adams (1966), & Anderson’s-Black Rock (1969)); Patentable subject matter (Flook (1978), Charkabarty (1980), Diehr (1980)); Prosecution history estoppel (Warner-Jenkinson (1995) & Festo (2000)).
The case also represents a growing trend of pitting patient advocates (who want cheaper access to medicine) against innovators (who create better medicine).
Notes & Documents:
Claim 1 of the ‘302 patent: A method of optimizing therapeutic efficacy for treatment of an immune-mediated gastrointestinal disorder, comprising:
(a) administering a drug providing 6-thioguanine to a subject having said immune-mediated gastrointestinal disorder; and
(b) determining a level of 6-thioguanine or 6-methyl-mercaptopurine in said subject having said immune-mediated gastrointestinal disorder,
wherein a level of 6-thioguanine less than about 230 pmol per 8108 red blood cells indicates a need to increase the amount of said drug subsequently administered to said subject and
wherein a level of 6-thioguanine greater than about 400 pmol per 8108 red blood cells or a level of 6-methyl-mercaptopurine greater than about 7000 pmol per 8108 red blood cells indicates a need to decrease the amount of said drug subsequently administered to said subject.
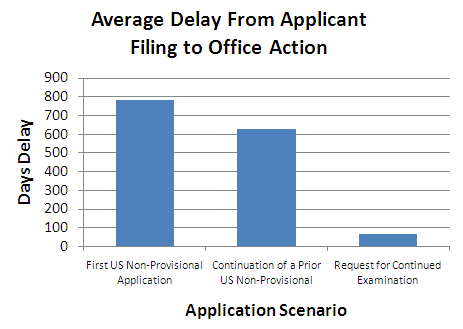
 Following the Sunstein-Thayler nudge theory, the new PTO administration is taking small non-rule-based steps to discourage the filing of Requests for Continued Examination (RCEs). RCE’s are already fairly expensive – $810. That outlay is relatively low compared with the cost of preparing and filing an appeal – especially when it appears that an applicant’s arguments have not been fully considered or when the applicant perceives the opportunity for an amendment that would lead to allowance.
Following the Sunstein-Thayler nudge theory, the new PTO administration is taking small non-rule-based steps to discourage the filing of Requests for Continued Examination (RCEs). RCE’s are already fairly expensive – $810. That outlay is relatively low compared with the cost of preparing and filing an appeal – especially when it appears that an applicant’s arguments have not been fully considered or when the applicant perceives the opportunity for an amendment that would lead to allowance.