Guest post by Professors Mark A. Lemley and Lisa Larrimore Ouellette of Stanford Law School.
Two of the most controversial patent law changes of the past year have involved obviousness-type double patenting, which allows applicants to patent obvious variants of their earlier patents by disclaiming the extra term of the later-expiring patent. First, the Federal Circuit held in In re Cellect that patents tied by double patenting must expire on the same day even if one of the patents has received a patent term adjustment (PTA) giving it a more-than-twenty-year term. The en banc denial and pending cert petition in Cellect have attracted substantial interest from amici and commenters. Second, the USPTO proposed that terminal disclaimers include a commitment to not enforce the patent if any claims in the patent to which it is tied are found to be obvious or not novel. Early commentary on the proposal was “overwhelmingly negative.”
In our new draft article, Fixing Double Patenting, we argue that this outcry is unwarranted. Both Cellect and the USPTO’s proposed regulations are sensible, modest steps to limit the abuses associated with double patenting. These efforts will enable inventors and the USPTO to focus on what should be important—inventing and patenting new inventions rather than multiplying patents on trivial variants of old inventions.
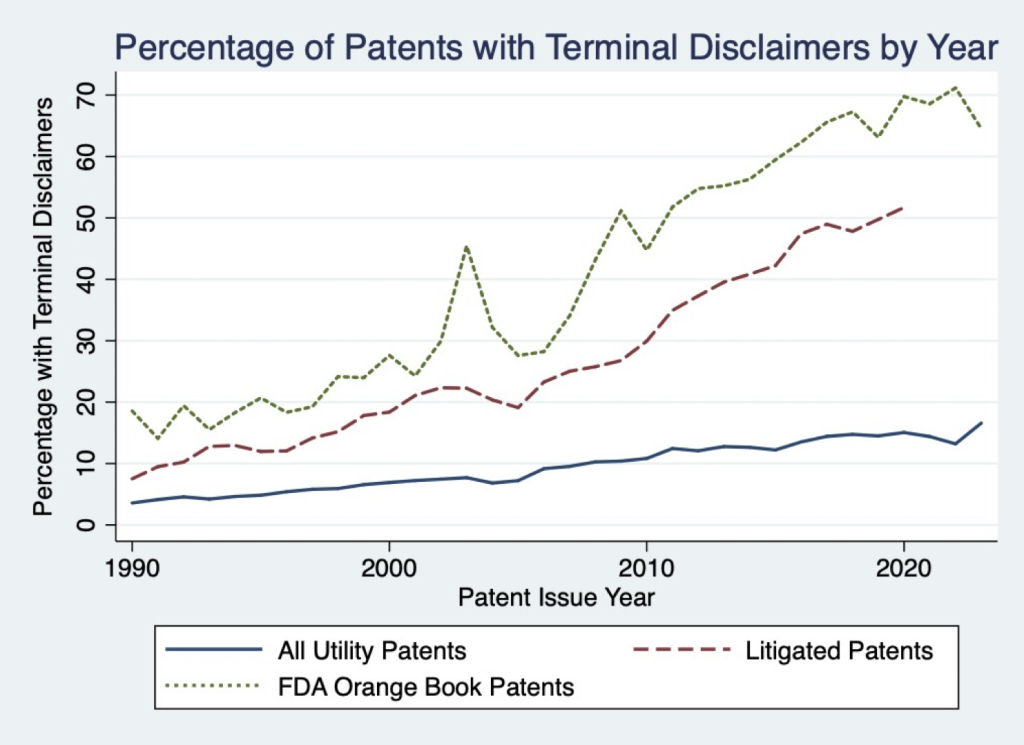
Our article provides comprehensive new evidence on the surprising extent of double patenting, particularly for important categories of patents. As illustrated in the figure, terminal disclaimers are now being filed for over 15% of all patents, over 50% of litigated patents, and well over 60% of patents covering FDA-approved drugs.
Terminal disclaimers are designed to address the concern with improper extension of patent term, but they have not prevented all double patenting gamesmanship. One policy problem stems from what Lemley and Reinecke dub “our more-than-twenty-year patent term.” Most patents get at least some PTA, and those that do get more than a year on average. If an inventor files multiple patents on obvious variants of the same invention, it is thus statistically likely that some of them will start their term early, while others will receive substantial additional term at the end of the patent life.
The Federal Circuit’s Cellect decision addresses this concern. Under the new rule, a patent owner is still entitled to PTA for one patent to compensate for any USPTO delay—they simply must disclaim additional extra term beyond the PTA collected on the first patent. That seems entirely consistent with the purpose of the terminal disclaimer. Many patent owners have already chosen to file terminal disclaimers on patents that would otherwise have significant amounts of PTA—of all patents with terminal disclaimers, 41% have term extensions, with an average extension of one year. And other patentees can correct their patents by adding terminal disclaimers any time before the first patent expires.
A separate policy problem is that the proliferation of multiple patents on trivial variants of the same invention generates transaction costs even if those patents expire simultaneously. The increase in double patenting places an increasing burden on examiners, which “negatively impacts the USPTO’s workload and docketing practices.” When these under-resourced examiners mistakenly grant invalid patents, it imposes additional costs in both money and time on innovators and entrepreneurs who want to evaluate, license, or litigate existing patents.
The USPTO’s proposed revision of its terminal disclaimer regulations was motivated by this concern. The proposal wouldn’t affect any existing patents. But new terminal disclaimers would have to affirm that the patent with the disclaimer won’t be enforced if the patent to which it is tied has any claim invalidated as anticipated or obvious. In our article, we explain why it is difficult to predict how this rule would affect patenting behavior or prosecution costs, including for small entities. Overall, it is likely to not only reduce the social costs of double patents that are issued, but also to encourage more applicants to prepare a compact application with the most relevant claims from the outset—as opposed to taking a many-bites-at-the-apple approach of seeking a sprawling family of overlapping patents. We also discuss the pros and cons of variations of the proposal, such as amending the rule to a claim-specific approach under which patentees would expressly identify the overlapping claims that triggered the disclaimer.
Critics of Cellect and the USPTO’s proposed regulatory revision have largely focused on the cost to innovation, such as the threat of “serious harm to America’s innovation economy.” But there is no evidence to support this argument. Critics have made no showing of any innovation benefit of the current rules compared with greater restrictions on double patenting, and the transaction costs created by the proliferation of patents on trivial variants of old inventions are a presumptive harm. We thus think that reducing abuses associated with double patenting makes good policy sense. Indeed, as we explain in more detail in our article, the USPTO and the courts could do more to restrict double patenting, and we evaluate additional options for reform.
Portions of this post are adapted from our article, which is available on SSRN.
“When these under-resourced examiners mistakenly grant invalid patents, it imposes additional costs in both money and time on innovators and entrepreneurs who want to evaluate, license, or litigate existing patents.”
This is a ridiculous justification and it is not even logical. I’ve done litigation, patent prosecution, and licensing for 20 years. When we file a continuation, the examiner already knows the invention and the disclosure but gets the same amount of time to examine the continuation application. Get it? The examiners have more time to examine continuations.
Continuations are what we have used to fight people like Lemley who has advocated for creating fragmented patent law where patent rights are reduced at every opportunity. With continuations we can file new claims when the CAFC holds that “a processor” does not cover two or more processors. Or a thousand other holdings of the CAFC that limit the patent right.
The fact is that Mark Lemley does not think that patents should exist for anything but a small fraction of innovation and rather than making his arguments to Congress he has made a living of attacking patents through judicial activism. It has decimated patent law where there is no predictability and no consistency in patent law. But don’t think that Lemley is so powerful as he is back by Google and others. The fact is patents are an integral part of the innovation system and it is being broken and the fact that we now behind in many technologies to China and other can be directly traced to Lemley’s assault (well-funded by corporations) on the patent system.
Continuations probably account for about half the value of patents right now as they have been used as work arounds in the face of d#ath squads, anti-patent judicial activist CAFC judges, the Scotus being duped into believing patents need to be weakened, and so forth.
Additionally, the TD is merely a convenience to save the applicant money and the examiner time. Right now, unless you are in the pharmaceutical industry, you generally will file a TD rather than fight a obviousness type double patenting rejection. If these rules are adopted, this will change and patents will instantly drop in value another 25%-50%.
This is just another Lemley anti-patent judicial activist paper.
+1
+1
Lemley? Not gonna bother to read it.
Meh. Better to read and understand what he thinks that he understands.
Is it only me or is it bothersome that the author cites himself when trying to support his argument….
“One policy problem stems from what Lemley and Reinecke dub “our more-than-twenty-year patent term.”
Instead of citing, why not just say “as the authors have previously proposed..” The citation appears as if the author is trying to elevate the impact of his argument.
+1
Because he has lots of mental issues from destroying our patent system for money.
It is not lost on me that Professors Lemley and Ouellette had Jason turn on the comments for this. Most of Jason’s guest author articles opt for no comments.
Perhaps because for those articles that comments are turned off, there is an immediate reduction of impact for those desired narratives.
Apologies if this question is addressed above, but does the data reflect when the USPTO reinterpreted the MPEP to require TDs to be filed, even in the case of the rejection being provisional?
The 20+ year term in the Lemley/Reinecke article title is misleading. Is the paper supposed to be unbiased research or advocacy work? A proper label would help me gauge my response accordingly.
Nevertheless, I read the article, which is more balanced, and I am treating it as research as opposed to advocacy work at this juncture. That said, the recommendation to make software patents harder to get to free up resources in other areas of the USPTO is bizarre and not well-formed. For example, software examiners do not work on pharma cases. Moreover, practically, software patents cover a lot of overhead cost for the USPTO, which is a for-profit enterprise within the federal government. it swings to a loss with a large drop off in software-related patents.
In my data sets, the average prosecution window for a parent patent, tied to its earliest effective filing date as of today, is about 20.80 years for all utility grants without terminal disclaimers on or after 1/1/2005. I do not account for leap years, which is an insignificant transgression.
The average protectable term for the same data set, which starts on the grant date, is about 17.43 years for parent utility patent assets lacking terminal disclaimers.
If you include continuations/CIPs/divisionals/etc. in that data set, the term drops to 16.89 years.
If you include assets with terminal disclaimers, the term drops to 16.53 years.
The data refutes the 20+ year protectable term claim in the article’s heading.
This is an easy question to test regarding the 20+-year term without a lot of hypothesis testing: take a filed application yet to grant to court and sue for damages. Anyone want to take bets on whether the case survives summary judgment?
I think their point is that the total term for each invention exceeds 20 years (or 17 years, which was the aim of the URAA amendments) because family members being in force does not stop PTA from accruing, so you earn PTA on the back end while having a different patent in force during the time of delay anyway.
DC seemed to indicate though that parent patents are often the ones accruing the PTA, and then having their term cut short by a later-issued, earlier-expiring child, and I even agree that’s kind of weird.
“that parent patents are often the ones accruing the PTA, and then having their term cut short by a later-issued, earlier-expiring child..”
Happens all the time in Pharma. Examiner takes an enormously long time to grant a compound patent. Say 3+ year of PTA. File a CON to the use of that compound in disease X, patent sails through in record time because compound is novel so any use of the compound is also novel. Easy points for the Examiner. Second patent grants with no added term.
Result = Parent compound parent term (with PTA) cut short by a method of treating child patent (No PTA).
What can be helpful is that the pharma arts are highly-unpredictable, and absent admissions by applicant in the spec or clearly present in the prior art, the activity of a given new compound can often be found unpredictable and hence non-obvious. This can enable one to file the new use application as a stand-alone case without claiming priority to the parent.
Highly unpredictable?
I thought that those were the “Grown-up Arts”
According to the FDA’s own statistics, 91% of all drugs prescribed in 2022 were generic. There is not a drug patent crisis. The fees and the proposed TD rule are both designed to curtail or eliminate current continuation practice in the US and make all patents easier to invalidate.
Let’s leave US patent law alone and try to fix any perceived problems by focusing on the behavior of patent litigants and their counsel. Clients and their counsel should both have some skin the litigation game. Lawyers: ask your clients to pay billed hours + a percentage of any judgement if an asserted patent claim is valid and infringed. Clients: find a lawyer that will refund 35% of hours billed if the claims are found to be obvious. This will separate the wheat from the chaff in short order.
Maybe also put some more teeth into Rule 11, such as requiring the bereaved patentee to conduct a (non-discoverable) comprehensive patent search prior to patent litigation. See, for example, link to patentlyo.com For the communists that want to ban continuation practice outright, these search results will necessarily be disclosed in any pending US continuations and can be used by competitors to invalidate all similar “obvious-type” claims.
These suggestions help weed out the “Sweet. $5M in billable litigation hours, win or lose, will allow me buy a second vacation home” cases from the cases with actual merit. Meritless litigation is what GARMIN and MERCEDES are really complaining about. Not drug prices.
The propsed rule around invalidating continuations is based on the incorrecct assumption that a) all continuations are obvious in light of their parent and that b) because the parent (or only one claim!) is invalid, all claims of all continuations must also be invalid.
USPTO current practice means that almost all continuations are subject to a terminal disclaimer, even if they claim something completely different. Applicants don’t contest TD requirements because of the cost and the relatively low impact of a TD. There’s no gurantee that any continuation is obvious in light of the parent based on the existence of a TD.
It’s also a dangerous assumption to state that all child claims are invalid just because one claim of a parent is. Continuations may have much narrower claims or are often filed for very different aspects of an invention, effectively a divisional by another name. Many applicants will pursue a strategy of getting one invention granted, and then pursuing other inventions via continuations. If the proposal goes through then you would expect to see many more divisionals filed.
I’ve seen in multiple articles on the topic, including the USPTO proposal, that TDs are ‘voluntary’. (‘chosen’ in the present article). This is blatantly untrue and I don’t understand where these writers get the idea that they are voluntary. TDs are always imposed by USPTO examiners. I can’t think of a good reason to voluntarily file a TD, that is apart from heading off an office action in advance.
Really good points.
“A separate policy problem is that the proliferation of multiple patents on trivial variants of the same invention generates transaction costs even if those patents expire simultaneously. The increase in double patenting places an increasing burden on examiners, which “negatively impacts the USPTO’s workload and docketing practices.””
This is hilarious. Absolutely no examiner complains about this. Nor would the USPTO as this helps them with pendency (quick turnaround on 20% of patents) and funding. Mark Lemley doesn’t know the first thing about prosecution.
Yup. This is not only hilarious it is the opposite. The examiner gets the same amount of time for a continuation as the original application. And the examiner is already familiar with the invention.
Every examiner I’ve ever worked with says they love continuations.
It’s been clear since he wrote that article with J. Moore many years ago that Prof. Lemley has no understanding of the examination process and the motivations of examiners. He thinks he does, but he does not.
Hard to believe that this was not an intentional “mistake” given what I’ve seen in other Lemley papers.
“Under the new rule, a patent owner is still entitled to PTA for one patent to compensate for any USPTO delay—they simply must disclaim ADDITIONAL extra term beyond the PTA collected on the first patent.”
This seems intentionally misleading. I assume that Lemley and Ouellette understand that a later-filed, earlier-expiring patent can be used as an OTDP reference against an earlier-filed, later-expiring patent. In practice, the existence of ANY subsequent patent in a given family that can be potentially used as an OTDP reference against the first patent in the family will put PTA of the first patent at serious risk. Indeed, the first patent will often have PTA while a later continuation will not. These are the situations most practitioners are actually worried about following Cellect.
Perhaps Lemley and Ouellette should re-read Gilead v. Natco from 10 years ago in the context of Cellect and revise the above statement accordingly.
+1
The discussion assumes that terminal disclaimers are filed for applications that were properly rejected for double patenting. I challenge this assumption. My clients nearly allows want to minimize cost and save time by filing the terminal disclaimer regardless of the merits of the underlying rejection. If they have two patents that expire at the same time and no intention of separating ownership, the cost of argument simply isn’t justified. As the rules change, this calculation will change with it.
Potentially worse than the actual costs (USPTO and legal fees), actually arguing that improper OTDP rejection may create additional estoppel.
Yup.
Changes are NOT needed in the current terminal disclaimer practice. Certainly not changes along the lines of this proposed rule. There would be a significant adverse economic impact to small entities if the rule is adopted, as proposed.
Terminal disclaimers are a vital component in the practice of using continuation applications to obtain claims which define the claimed subject matter in a different way – and often in a narrower way – than existing claims in a case from which it claims priority.
I often represent small companies who are working on limited budgets while developing new technologies which have not been fully tested for a variety of embodiments. In such cases, the risk management versus budgetary constraints evaluation often results in the filing of applications with broad disclosures but with limited initial claim sets, often narrowly directed at a current prototype or at a specific initial market target. If a parent application is granted, a continuation application is then filed, and modified claims are filed to more squarely address another target market or specific technological application, or to more broadly cover certain aspects of the innovation.
Often, and especially if international applications are filed, the the universe of known material prior art expands over time. The US Patent Office often does not uncover prior art patents found by other patent offices. The authors recognize this problem, but the proposed fix does not address the problem. It merely buries the problem, with the burden falling on the patent owner.
Inevitably the US Patent Office additionally does not uncover much of the non-patent prior art presented by competitors that have opposed a patent grant, for example in Europe or Japan. (Importantly, those countries have a much better mechanism than does the US for amendment of claims during such opposition proceedings, in order to clearly avoid prior art yet result in allowance of valuable patent claims.) It is not uncommon for the additional material prior art which becomes known after issue of a parent US case to result in lack of novelty of one or more claims in the parent case, or to make the question of non-obviousness of one or more claims a close call.
Consequently, the availability of a VIABLE continuation application – and resulting issued patent – with full benefit of a terminal disclaimer under current practice, is crucial for providing a vehicle to present clearly allowable claims in full view of all subsequent prior art uncovered. The proposed KILL SWITCH which would result via adoption of a final rule in the form of the currently proposed rule would render any continuation claims invalid and unenforceable IF ANY of the parent case claims are found invalid. Such a drastic change in US patent practice would totally disable the current ability of small entities to ultimately obtain a valid patent in view an expanded prior art universe. Perversely, the proposed rule may well be construed as an attempt by the US Patent Office to “bury mistakes” made in patent issue due to deficiencies in current search capabilities at the time of examination. Small companies and independent inventors should not bear the burden of the USPTO’s limited patent searching capabilities.
Moreover, it is entirely unreasonable to change the current practice of obtaining amended claim sets via way of continuation applications. To do so would inevitably force patent owners to seek revisions to the claims in their existing US patents via ex parte reexamination procedures, in order to strengthen their patent claims against invalidity challenges.
Great points! Or later CAFC opinions like the “a processor” doesn’t cover two accused processors.
This statement is untrue:
“Under the new rule, a patent owner is still entitled to PTA for one patent to compensate for any USPTO delay—they simply must disclaim additional extra term beyond the PTA collected on the first patent.”
The PTO is rejecting earlier-issued patents that have PTA as obvious over much later issued patents. See e.g., ctrl. no. 90/019,130. This issue applies to one patent in Cellect, but it’s been lost in the shuffle, and no one has focused that specific issue.
The PTO is rejecting any patent that expires later than a double patent. According to the PTO, issue date is irrelevant. Therefore, there are also less than 20-year patents under the new regime.
Stated differently, there are inventions that do not receive the “guaranteed” 17-year effective term due to USPTO delays.
What do you propose is the fairest way to resolve the issues of obviousness type double patenting and appropriate patent term? What is the legislation you would like to see?
Why do you think that new legislation is needed?
Ban double patenting!!! I thought it was already?
What about the judicially created doctrine of obviousness-type double patenting? Isn’t that something different?
Or….. does that spoil the impact of the misleading title?
Eminent author, indeed, but I suggest prosecuting a few US patent cases to see how this works in the real world. The view is different from that of an iv(or)y tower.
All of which ignores the question of whether the PTO has any authority to create such a legistative-type rule. Multiple comment letters, including one from former Director Iancu and other former PTO officials, argue that the PTO has no such authority.
Not surprisingly, sampling the comment letters filed so far, they almost uniformly appear to be telling the PTO to drop this proposed rule.