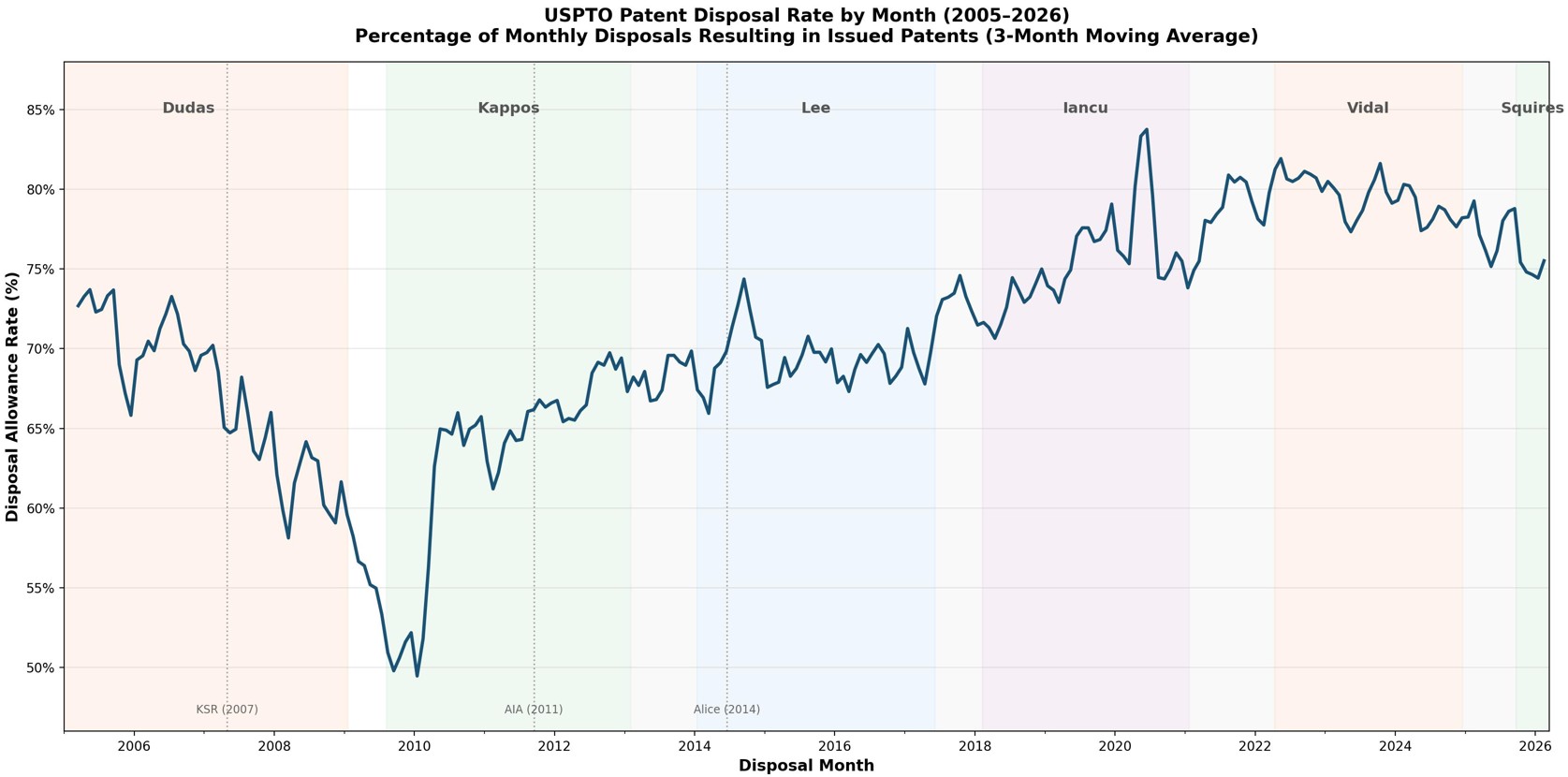
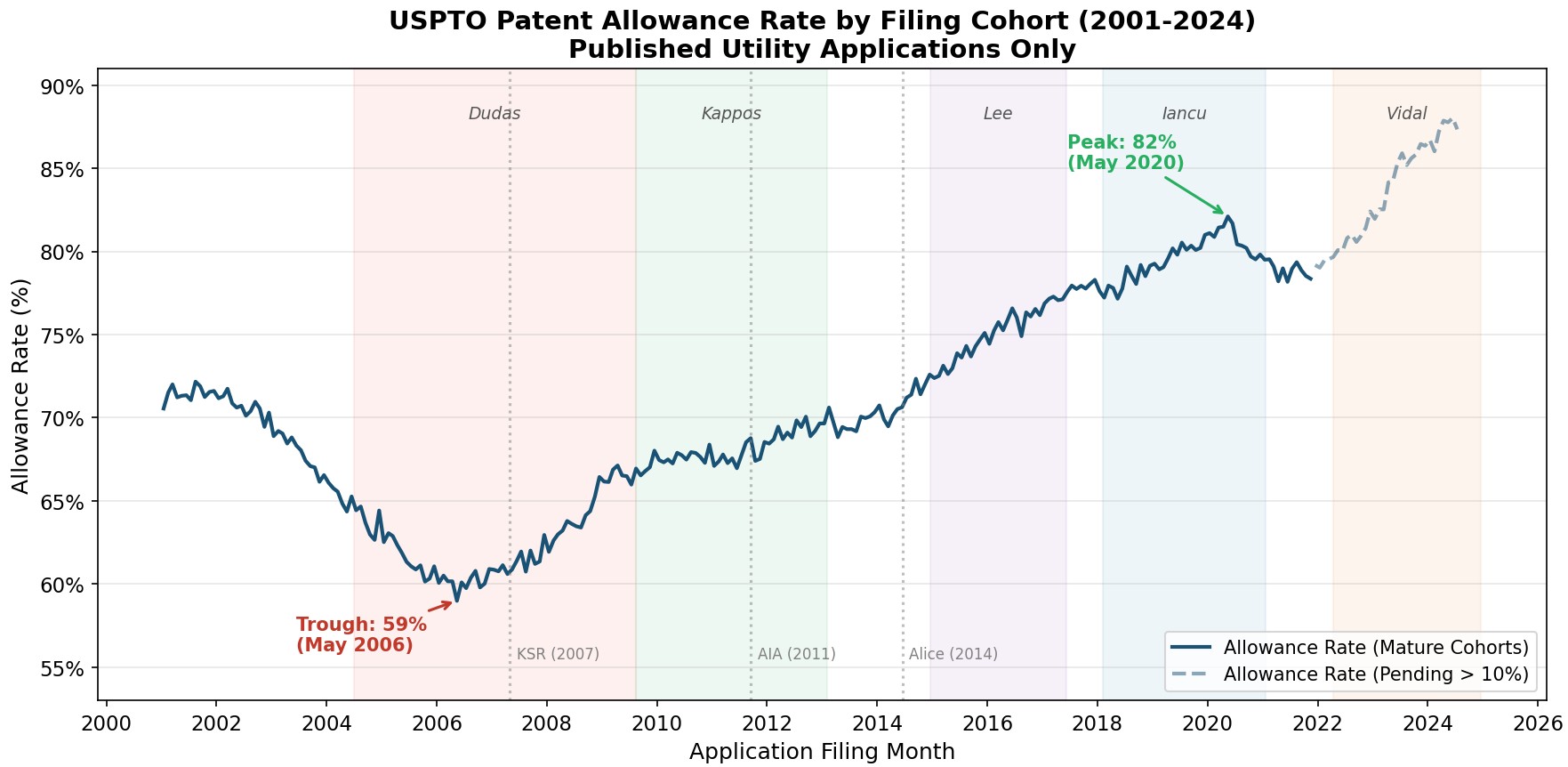
by Dennis Crouch
The America Invents Act requires that every IPR petition “identif[y] all real parties in interest.” 35 U.S.C. § 312(a)(2). The statute uses mandatory language: the petition “may be considered only if” this condition is satisfied. On its face, the provision creates an obligation directed at petitioners and a corresponding entitlement for patent owners and the public. But does the failure to comply with that requirement give a patent owner the kind of concrete injury that Article III demands? The Federal Circuit says no. Dolby Laboratories is now asking the Supreme Court to disagree.
The petition for certiorari in Dolby Laboratories Licensing Corp. v. Unified Patents, LLC, No. 25-1011 (U.S., filed Feb. 20, 2026), challenges the Federal Circuit’s June 2025 decision holding that the AIA does not create an “informational right” for patent owners to know the identities of all real parties in interest. Dolby Laboratories Licensing Corp. v. Unified Patents, LLC, 138 F.4th 1363 (Fed. Cir. 2025).
Dolby had won the IPR, but still wanted to know the identity of the people behind the patent challenge. But, the Federal Circuit dismissed Dolby’s for lack of Article III standing, despite statutory text that conditions the consideration of an IPR petition on full RPI disclosure. The case raises two questions:
- Whether a patent owner suffers an injury in fact when the Board refuses to enforce the RPI identification requirement, and
- Whether 35 U.S.C. § 314(d) bars judicial review of the Board’s RPI determinations in its final written decision.
These questions fit within an an area known as “informational standing doctrine” often tied to FOIA and Sunshine law requests. The Supreme Court has recognized that the denial of information to which a plaintiff is legally entitled can constitute a cognizable injury in fact, at least in certain statutory contexts. See, TransUnion LLC v. Ramirez, 594 U.S. 413 (2021). In Public Citizen v. Department of Justice, 491 U.S. 440 (1989), the Court held that the denial of access to records from an advisory committee constituted injury in fact because the Federal Advisory Committee Act (FACA) requires that advisory committee records be made publicly available. In FEC v. Akins, 524 U.S. 11 (1998), the Court found standing where the Federal Election Campaign Act (FECA) required political committees to disclose detailed reports of donors and expenditures, and FECA provided an express statutory cause of action for any party who believed a violation had occurred.
The Federal Circuit in Dolby distinguished these sunshine-law contexts and the AIA. Chief Judge Moore’s opinion reasoned that the purpose of FACA and FECA is to grant public access to information, while the purpose of the AIA is “to establish a more efficient and streamlined patent system that will improve patent quality and limit unnecessary and counterproductive litigation costs.” quoting Thryv, Inc. v. Click-To-Call Techs., LP, 590 U.S. 45 (2020). Because the AIA’s purpose is efficiency rather than transparency, the court concluded, § 312(a)(2) does not create an “informational right” at all. And even if patent owners have some right to have RPI disputes adjudicated, “such a right only arises in the context of IPR proceedings; there is no freestanding right to that information.”
In these cases, the patent owner wants that information for a few reasons:
- Enforcing the statutory estoppel provisions of 35 U.S.C. § 315(e), which attach to “the petitioner” and “the real party in interest or privy of the petitioner.”
- Administer the one-year time bar of § 315(b), which prevents institution when a real party in interest was served with an infringement complaint more than a year before the petition was filed.
- Ensuring PTAB judges avoid conflicts of interest, which they cannot effectively do so if the actual parties behind a petition are unknown.
The Federal Circuit dismissed each of these as too speculative on the facts of this case.
The § 314(d) Problem. The Federal Circuit also held that, even if patent owners have some right to RPI adjudication, § 314(d) bars judicial review of the Board’s decisions concerning the § 312(a)(2) RPI requirement. The basic conclusion here is that RPI identification is part of the institution decision, and institution decisions are not subject to appeal. Dolby’s petition invokes SAS Institute, Inc. v. Iancu, 584 U.S. 357 (2018), as directly contrary. In SAS, the Supreme Court confronted the Director’s practice of “partial institution” and found that appealable. § 314(d) as precluding review of the Director’s “initial determination” under § 314(a), but does not bar review of whether the agency exceeded its statutory bounds in conducting the proceeding.